Cellular analysis of bronchoalveolar lavage fluid in various lung lesions
Abstract
Introduction: Bronchoalveolar lavage (BAL) is a standard tool in the diagnosis of lung diseases. Cellular analysis of BAL fluid when deviates from normal are indicative of an inflammatory/infiltrative process. This study involves an analysis of BAL fluid performed manually and also using an automated analyzer which offers a rapid and reliable method for analysis of unprocessed BAL fluid.
Aims and Objectives: To utilize the cellular analysis of bronchoalveolar lavage (BAL) fluid in the diagnosis of various lung diseases.
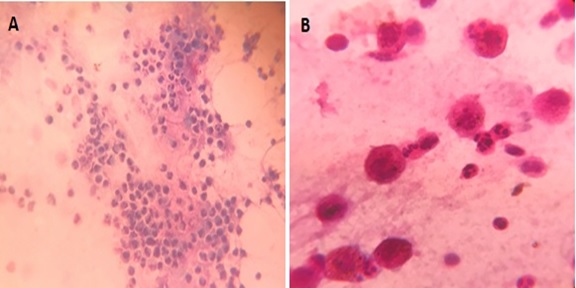
Materials and Methods: 37 patients with pulmonary diseases of varying etiologies were included in this study. Fiberoptic bronchoscopy with BAL was performed. BAL fluid was run in SYSMEX XNL/350 six-part analyzer which gave total and differential cell count (mononuclear and polymorphonuclear cells). The sample was also centrifuged, sediment smears prepared and differential cell counts were determined.
Results: Total WBC count ranged from 31 cells/µl to 94 cells/µl with the maximum increase seen in asthma. Mononuclear cells were increased in the majority of cases, being 84% in pneumoconiosis with the predominance of macrophages. An increase in lymphocytes was seen in Non-Specific Interstitial Pneumonia (45.3%) and Bronchiolitis Obliterans Organising Pneumonia (35.3%), neutrophils in Usual Interstitial Pneumonia (26.2%) and eosinophils in Chronic Eosinophilic Pneumonia (55.5%) and asthma (8.4%). There were 58% mononuclear cells and 42% polymorphonuclear cells in tuberculosis. There were two cases of adenocarcinoma and one case of squamous cell carcinoma in the present study.
Conclusion: BAL cell differentials when combined with additional clinical and radiographic information help secure a confident diagnosis in lung diseases and help clinicians to make therapeutic decisions.
Downloads
References
Meyer K C. The role of bronchoalveolar lavage in interstitial lung disease. Clin Chest Med. 2004;25(4):637-649. doi: https://doi.org/10.1016/j.ccm.2004.08.001.
Kilinc G, Kolsuk E A. The role of bronchoalveolar lavage in diffuse parenchymal lung diseases. Curr Opin Pulm Med. 2005;11(5):417-421. doi: https://doi.org/10.1097/01.mcp.0000175522.49353.e1
Pacinin IFM, Camargos PAM, Marguet C. Cell profile of BAL fluid in children and adolescents with and without lung disease. J Bras Pneumol. 2010;36(3):372-391. doi: https://doi.org/10.1590/S1806-37132010000300016.
Welker L, Jorres R A, Costabel U, Magnussen H. Predictive value of BAL cell differentials in the diagnosis of interstitial lung diseases. Eur Respir J. 2004;24:1000-1006. doi: https://doi.org/10.1183/09031936.04.00101303.
Meyer K C. The clinical utility of bronchoalveolar lavage in interstitial lung disease- is it really useful? Expert Rev Respir Med. 2014;8(2):133-135. doi: https://doi.org/10.1586/17476348.2014.879827.
Heaney L G, McKirgan J, Stanford C F, Ennis M. Electronic cell counting to measure total cell numbers in bronchoalveolar lavage fluid. Eur Respir J. 1994;7:1527-1531. doi: https://doi.org/10.1183/09031936.94.07081527.
Natiello M, Kelly G, Lamca J, Zelmanovic D, Chapman R W, Phillips J E. Manual and automated leucocyte differentiation in bronchoalveolar lavage fluids from rodent models of pulmonary inflammation. Comp Clin Pathol. 2009;18:101-111. doi: https://doi.org/10.1007/s00580-008-0772-9.
Lee W, Chung W S, Hong K S, Huh J. Clinical usefulness of bronchoalveolar lavage cellular analysis and lymphocyte subsets in diffuse interstitial lung diseases. Ann Lab Med. 2015;35(2):220-225. doi: https://doi.org/10.3343/alm.2015.35.2.220.
Nagai S, Kitaichi M, Itoh H, Nishimura K, Izumi T, Colby T V. Idiopathic nonspecific interstitial pneumonia/fibrosis: comparison with idiopathic pulmonary fibrosis and BOOP. Eur Respir J. 1998;12:1010-1019. doi: https://doi.org/10.1183/09031936.98.12051010.
Ozaki T, Nakahira S, Tani K, Ogushi F, Yasuoka S, Ogura T. Differential cell analysis in bronchoalveolar lavage fluid from pulmonary lesions of patients with tuberculosis. Chest. 1992;102(1):54-59. doi: https://doi.org/10.1378/chest.102.1.54.
Cordeiro C R, Jones J C, Alfaro T, Ferreira A J. Bronchoalveolar lavage in occupational lung diseases. Semin RespirCrit Care Med. 2007;28(5):504-513. doi: https://doi.org/10.1055/s-2007-991523.
Kayacan O, Beder S, Karnak D. Cellular profile of bronchoalveolar lavage fluid in Turkish miners. Postgrad Med J. 2003;79(935):527-530. doi: http://dx.doi.org/10.1136/pmj.79.935.527.
Wilson J W, Djukanovic R, Howarth P H, Holgate S T. Lymphocyte activation in bronchoalveolar lavage and peripheral blood in atopic asthma. Am Rev Respir Dis. 1992;145(4):958-960. doi: https://doi.org/10.1164/ajrccm/145.4_Pt_1.958.
Sampsonas F, Kontoyiannis D P, Dickey B F, Evans S E. Performance of a standardized Bronchoalveolar lavage protocol in a comprehensive cancer center. Cancer. 2011;117(15):3424-3433. doi: https://doi.org/10.1002/cncr.25905.
Lee JY, Park HJ, Kim YK, Yu S, Chong YP, Kim SH, et al. Cellular profiles of bronchoalveolar lavage fluid and their prognostic significance for non-HIV-infected patients with Pneumocystis jirovecii pneumonia. J Clin Microbiol. 2015;53(4):1310-1316. doi: https://doi.org/10.1128/JCM.03494-14.
Copyright (c) 2020 Author (s). Published by Siddharth Health Research and Social Welfare Society

This work is licensed under a Creative Commons Attribution 4.0 International License.


 OAI - Open Archives Initiative
OAI - Open Archives Initiative


