Significance of histologic grading using visual analogue scale in chronic gastritis
Abstract
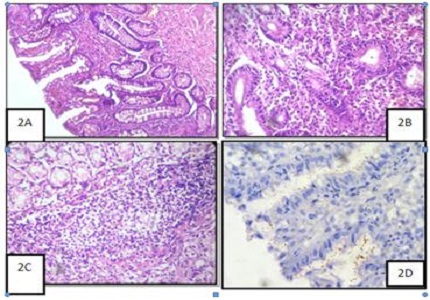
Introduction: Helicobacter Pylori and its association with carcinogenesis has significantly changed the field, establishing new guidelines for the detection of it. Carcinogenicity of H. Pylori necessitates grading the variables with visual analogue scale in accordance to Sydney system classification. The objectives of this study is to assess chronic gastritis and grade with visual analogue scale of Sydney classification.
Methods: This is a prospective study of 3 years in which 50 gastric endoscopic mucosal biopsies were included in the study. Grading of the variables were done with reference to Sydney classification. Morphological changes in all cases were studied with special reference to atrophy, dysplasia, metaplasia, neutrophilic infiltration, lymphoid follicle formation, plasma cell infiltration & malignant changes along with identification of H. pylori.
Results: Of the 50 cases studied for gastritis, 20 cases (44.4%) had Chronic H. pyloric gastritis followed by 18 cases (38.6%) of chronic gastritis with nonspecific features. Evaluation for graded variables showed 41 cases of gastritis with chronic inflammation of which 22 cases showed H. Pylori positivity. Of the 20 cases of gastritis showed inflammatory activity, 11 showed H. Pylori positivity. H. pylori was seen in 21 cases which could be graded. Of the 15 cases of gastritis showing surface epithelial damage, 9 cases showed H. pylori positivity. Chronic inflammation was most commonly observed and 22 of the 41 cases with chronic inflammation showed H.pylori positivity. Presence of H.pylori was an useful indicator in grading of gastritis.
Conclusion: Visual analogue scale is an useful indicator to histologically grade chronic gastritis and detection of H. Pylori and helps in better evaluation of patient in preventing the further progression of the disease.
Downloads
References
2. Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996 Oct;20(10):1161-81. [PubMed]
3. Miszputen SJ. Guia de Gastroenterologia. 2a. ed. São Paulo: Manole, 2006.
4. Graham D. Tratado de Medicina Interna. São Paulo, Saunders Elsevier, 2005.
5. Adamu MA, Weck MN, Gao L, Brenner H. Incidence of chronic atrophic gastritis: systematic review and meta-analysis of follow-up studies. Eur J Epidemiol. 2010 Jul;25(7):439-48. doi: 10.1007/s10654-010-9482-0. Epub 2010 Jun 29.
6. Aydin O, Egilmez R, Karabacak T, Kanik A. Interobserver variation in histopathological assessment of Helicobacter pylori gastritis. World J Gastroenterol. 2003 Oct;9(10):2232-5.
7. Rautelin H, Blomberg B, Fredlund H, Järnerot G, Danielsson D. Incidence of Helicobacter pylori strains activating neutrophils in patients with peptic ulcer disease. Gut. 1993 May;34(5):599-603.
8. Zhuang Z, Wang S. Corrigendum to Histone Methyltransferase aflrmt A gene is involved in the morphogenesis, mycotoxin biosynthesis, and pathogenicity of Aspergillus flavus" [Toxicon 127 (2017, Mar 1) 112-121]. doi: 10.1016/j.toxicon.2017.01.013. Epub 2017 Jan 19. Toxicon. 2017 Aug;134:64. doi: 10.1016/j.toxicon.2017.05.026.
9. Park J, Kim MK, Park SM. Influence of Helicobacter pylori colonization on histological grading of chronic gastritis in Korean patients with peptic ulcer. Korean J Intern Med.1995;10(2):125-129.
10. Witteman EM, Mravunac M, Becx MJ, Hopman WP, Verschoor JS, Tytgat GN et al. Improvement of gastric inflammation and resolution of epithelial damage one year after eradication of Helicobacter pylori. JCP,1995; 48(3):250-256.
11. Rugge M, Correa P, Dixon MF, Fiocca R, Hattori T, Lechago J, Leandro G, Price AB, Sipponen P, Solcia E, Watanabe H, Genta RM. Gastric mucosal atrophy: interobserver consistency using new criteria for classification and grading. Aliment Pharmacol Ther. 2002 Jul;16(7):1249-59.
12. Rokkas T, Filipe MI, Sladen GE. Detection of an increased incidence of early gastric cancer in patients with intestinal metaplasia type III who are closely followed up. Gut. 1991 Oct; 32(10): 1110–1113.
13. Eidt S, Stolte M. Prevalence of lymphoid follicles and aggregates in Helicobacter pylori gastritis in antral and body mucosa. J Clin Pathol 1993;46:832–835.
14. Patwari AK, Anand VK, Malhotra V, Balani B, Gangil A, Jain A, Kapoor G. Brush cytology: an adjunct to diagnostic upper GI endoscopy. Indian J Pediatr. 2001 Jun;68(6):515-8.
15. Afzal S, Ahmad M, Mubarik A, Saaed F, Rafi S, Saleem N, Hussain A. Morphological Spectrum Of Gastric Lesions - Endoscopic Biopsy Findings. Pak Armed Forces Medical J 2006;2:1-9.
16. Misra V, Misra SP, Singh MK, Singh PA, Dwivedi M. Prevalence of H. pylori in patients with gastric cancer. Indian J Pathol Microbiol. 2007 Oct;50(4):702-7. [PubMed]
17. Pruthi S, Nirupama M, Chakraborti S. Evaluation of gastric biopsies in chronic gastritis: Grading of inflammation by Visual Analogue Scale. Med J DY Patil Univ 2014;7:463-7.
18. Dhakhwa R, Acharya IL, Shrestha HG, Joshi DM, Lama S, Lakhey M. Histopathologic study of chronic antral gastritis. J Nepal Health Res Counc. 2012 Jan;10(1):57-60.
19. Palaniappan V, Venkatraman Janarthanam and Swaminathan K. (2016). Histomorphological profile of Gastric antral mucosa in Helicobacter associated gastritis. Int. J. Curr. Res. Med. Sci. 2(4): 22-28.
20. Amarapurkar AD, Prabhu SR, Amarapurkar DN. Histological spectrum of lymphoid follicles and aggregates in Helicobacter pylori gastritis. Trop Gastroenterol. 1997 Jan-Mar;18(1):22-3.
21. Carrasco, G., & Corvalan, A. H. Helicobacter pylori-Induced Chronic Gastritis and Assessing Risks for Gastric Cancer. Gastroenterology Research and Practice, 2013, 393015.


 OAI - Open Archives Initiative
OAI - Open Archives Initiative


